Andalusite as Partial High-Alumina Bauxite Substitute: Enhance Copper Melting Furnace Refractories Greatly
In the steelmaking industry, furnace linings are subjected to severe chemical corrosion from high-temperature molten slag, often resulting in erosion, spalling, cracking, and material loss. Therefore, high-quality refractory materials with high refractoriness, high bulk density, low porosity, excellent slag resistance, and good thermal shock resistance are required.
Andalusite, a type of aluminosilicate mineral, has extremely abundant reserves in China. It typically occurs as columnar crystals with an approximately square cross-section. Its theoretical chemical composition is 63.1% Al₂O₃ and 36.9% SiO₂. Due to crystallization during ore formation, alteration, and other factors, it often contains impurities such as Ag, Fe, and Ti.
Within the temperature range of 1200–1600 °C, andalusite transforms into mullite, accompanied by a permanent volume expansion of 3%–5%. Mullite exhibits good corrosion resistance to copper slag, and the high-temperature transformation characteristics of andalusite can be utilized to improve the service performance of furnace lining materials.
In this paper, andalusite is used to partially replace high-alumina bauxite, and the effect of its addition on the properties of andalusite-based refractories is investigated.
01. Experimental Analysis
1.1 Raw Materials and Equipment
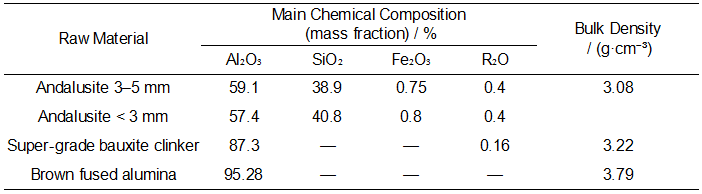
The main raw materials used in the experiment include super-grade bauxite clinker, andalusite, and brown fused alumina. Their main chemical compositions are listed in Table 1. Boric acid and dextrin are used as the fluxing agent and binder, respectively. The equipment used includes a high-temperature box-type resistance furnace, an electric heating blast drying oven, and a digital display hydraulic pressure testing machine.
1.2 Design of Experimental Scheme
During preparation, the main factors affecting the copper liquid corrosion resistance of andalusite-based refractories include raw material composition, particle size, sintering temperature, and the type of mineralizer. Considering the limited research time, this experiment mainly focuses on the influence of the andalusite content in the raw materials on the copper liquid corrosion resistance of the specimens.
Based on the principle that finer andalusite particles lead to a higher degree of mullitization, as well as the closest packing theory, four particle size fractions were adopted: 3–5 mm, 1–3 mm, 0–1 mm, and <0.088 mm. This allows large particles to form a framework during specimen preparation, while fine powder fills the voids in the framework to achieve closest packing. It also ensures that an appropriate amount of mullite can form after sintering.
It should be noted that boric acid was selected as the fluxing agent instead of multicomponent composite mineralizers. This promotes the formation of a three-layer corrosion-resistant structure in the sintered specimens, namely a damaged layer, a penetrated layer, and an unchanged layer, thereby slowing down the corrosion of the specimens by copper liquid.
The raw material composition, sintering temperature, and other parameters were determined through a literature review and preliminary experiments.
1.3 Experimental Method
On the premise that the total proportion of andalusite particles and super-grade bauxite clinker remained constant, seven groups of samples with different ratios were prepared by varying the amounts of andalusite particles and super-grade bauxite clinker. The raw material compositions are listed in Table 2.
All raw materials were dried and accurately weighed according to the designed ratios. After thorough mixing, the mixtures were pressed into specimens with dimensions of φ50 mm × 50 mm using a hydraulic press. A copper block (φ20 mm × 5 mm) was placed at the center of each specimen for the corrosion resistance test.
The specimens were dried at 110 °C for 24 h and then sintered in a resistance furnace at 1300 °C with a holding time of 3 h, followed by natural cooling. The corrosion morphology was observed, relevant performance parameters were measured, and XRD and SEM analyses were conducted on the sintered specimens.
02. Results and Discussion
2.1 Effect of Andalusite Addition on the Mechanical Properties of Specimens
After firing at 1300 °C for 3 h, the cold crushing strength, bulk density, apparent porosity, and linear change on firing of the specimens with different formulations (see Table 2) were measured, and the results are shown in Figure 1.
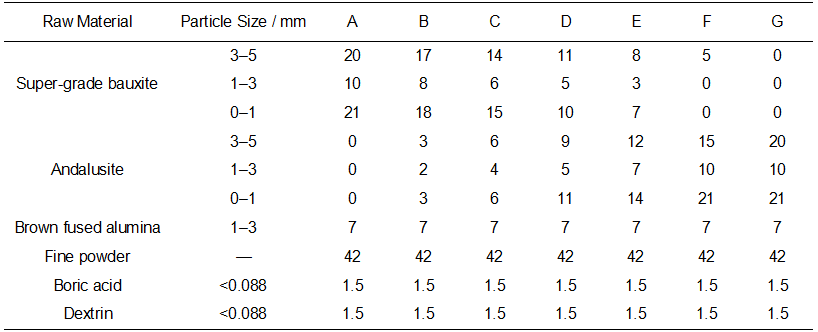
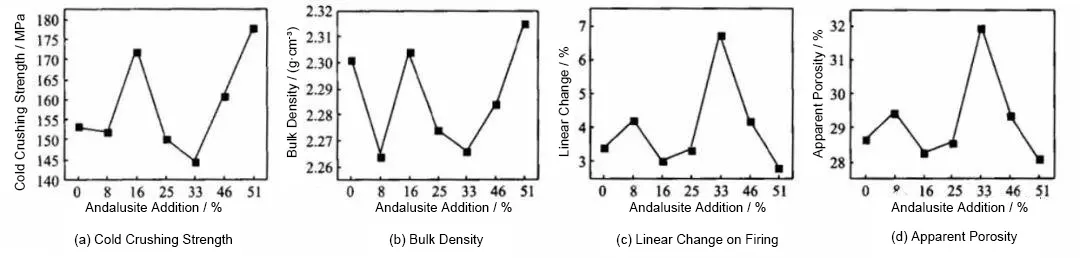
It can be seen from Figure 1a that the cold crushing strength of the specimens varies with the addition of andalusite. The strength reaches a first peak at an andalusite content of 16% and a second peak at 51%. This is mainly because, at a dosage of 16%, the primary mullitization reaction of andalusite (Equation 1) produces an appropriate amount of mullite. The SiO₂ liquid phase formed during the reaction cannot fully participate in the secondary mullitization reaction (Equation 2) and instead fills the spaces between the primary mullite particles. This not only suppresses the volume expansion caused by excessive mullite formation but also enhances the bonding between fine powder and particles through the acicular network structure of mullite, thereby improving the cold crushing strength.
3Al2O3· SiO2- 3Al2O3 · 2SiO2+SiO2 (1)
3Al2O3 + 2SiO2 - 3Al2O3 · 2SiO2(Mullite) (2)
At an andalusite content of 51%, the further increase in andalusite increases the amount of mullite formed during primary mullitization. The SiO₂ liquid phase produced by the reaction reacts with the added reactive SiO₂ micropowder to form additional mullite, resulting in a denser microstructure and leading to the second peak in cold crushing strength.
The variation trend of bulk density is opposite to that of apparent porosity and linear change on firing. Bulk density reaches its maximum values at andalusite contents of 16% and 51%.
At an andalusite content of 16%, as indicated by the analysis of cold crushing strength, primary mullitization of andalusite and filling by the SiO₂ liquid phase lead to a denser microstructure. When the andalusite content is increased to 51%, the higher addition promotes extensive mullite formation through both primary and secondary mullitization. The acicular network structure of mullite further develops and fills part of the open pores, resulting in an increase in bulk density.
2.2 Effect of Andalusite Addition on the Corrosion Resistance of Materials
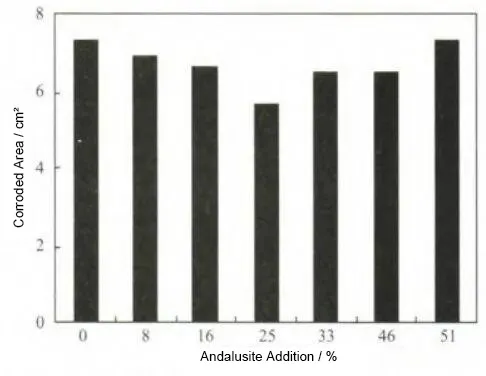
Figure 2 shows the variation in the corroded area of each specimen with different amounts of andalusite addition. It can be seen that, from formulation A to formulation G, as the andalusite content increases and the bauxite content in the raw materials decreases, the corroded area of the specimens first decreases and then increases. Among these, formulation D exhibits the best corrosion resistance.
This is because the surface of crushed and finely ground andalusite particles contains a large number of defects, and the crystal grains on the surface possess high energy, thus acting as active sites for mullite nucleation. Mullitization of andalusite first occurs on the crystal surface and then progresses inward from the surface to the interior. Limited by time and temperature, the andalusite inside the specimens cannot fully develop into coarse mullite crystals; instead, acicular mullite forms and interweaves into a network structure, thereby improving the corrosion resistance of the material.
Secondly, primary mullitization of the andalusite particles generates a considerable amount of SiO₂ glass phase, most of which remains in the capillary pores between mullite particles, filling the voids and increasing the density of the specimens. Finally, a small amount of SiO₂ liquid phase expelled to the surface further undergoes a secondary mullitization reaction with Al₂O₃, refining the acicular network structure.
Through comparison, it can be found that an appropriate addition of andalusite can effectively improve the copper liquid corrosion resistance of the specimens. When the andalusite content is 25% in formulation D, the above three mechanisms are maximized, thereby enhancing the corrosion resistance.
2.3 XRD Analysis
Figure 3 shows the XRD comparison patterns of three specimens with formulations A, D, and G.
It can be seen from Figure 3 that, with the increase in andalusite addition, the mullite content in the specimens reaches a maximum at 25% andalusite, while the mullite content in the other phases remains relatively low. This indicates that an appropriate ratio of andalusite to bauxite can promote the formation of a greater amount of mullite.
As the andalusite content continues to increase, the bauxite content in the raw materials decreases accordingly. As a result, the large amount of SiO₂ phase formed after primary mullitization lacks sufficient Al₂O₃ to participate in the secondary mullitization reaction.
The reduction in mullite content not only hinders the development of a complete acicular network structure but also causes a large amount of SiO₂ liquid phase to be expelled to the crystal surface, leading to detrimental effects.
Due to insufficient mullite formation, the fired specimens mainly exhibit volume shrinkage caused by high-temperature sintering, which also has a negative impact on apparent porosity, cold crushing strength, and bulk density.

2.4 SEM Analysis
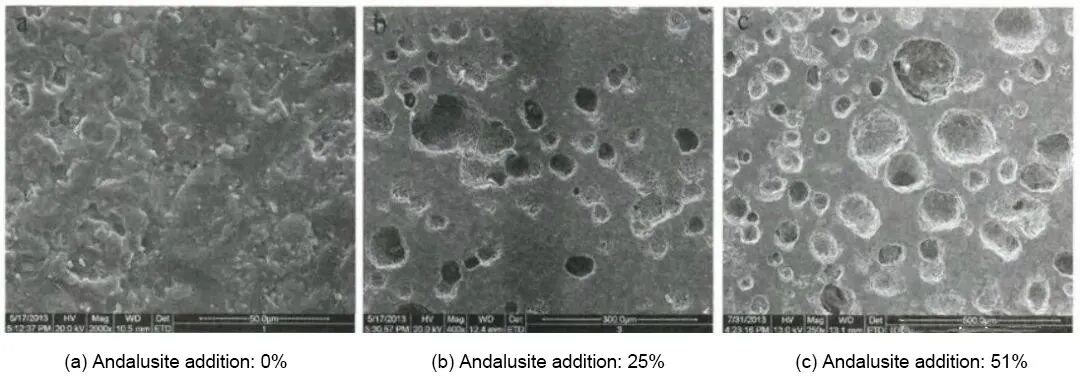
Figure 4 shows SEM images of the fracture surfaces of the specimens after high-temperature calcination. Figures 4a, 4b, and 4c present the fracture morphologies of specimens with andalusite additions of 0%, 25%, and 51%, respectively, after firing at 1300 °C for 3 h.
It can be seen from Figure 4a that, when no andalusite is added, no acicular mullite is observed in the specimen, which mainly consists of corundum and a glassy phase. In the absence of the network structure formed by acicular mullite, the internal structure of the specimen is loose, resulting in poor corrosion resistance.
Compared with Figure 4a, more pores can be observed in Figures 4b and 4c, which is attributed to the volume expansion associated with mullite formation. In Figure 4b, a network structure of acicular mullite can be seen surrounding numerous pores. This structure prevents the SiO₂ liquid phase formed during primary mullitization from being retained inside the matrix and instead drives it to the crystal surface, allowing it to fully react with Al₂O₃ and thereby promoting the secondary mullitization reaction.
This series of processes strengthens the bonding between the matrix and aggregate particles in the specimen, resulting in improved corrosion resistance and sintering performance.
03. Conclusions
(1) With the increase in andalusite content, both cold crushing strength and bulk density exhibit similar variation trends, showing two peaks. The variation trends of apparent porosity and linear change on firing are opposite.
(2) At an andalusite content of 16%, primary mullitization of andalusite and filling by the SiO₂ liquid phase result in a dense microstructure. At an andalusite content of 51%, the increased andalusite promotes the formation of a large amount of mullite through both primary and secondary mullitization, thereby strengthening the acicular network structure of mullite.
The specimens exhibit the best corrosion resistance at an andalusite content of 25%. At this content, primary mullitization of andalusite forms an acicular network structure, most of the SiO₂ liquid phase fills the pores, and the remaining SiO₂ liquid phase expelled to the crystal surface further undergoes secondary mullitization with Al₂O₃, refining the acicular mullite network.
Considering that this study mainly focuses on the corrosion resistance of the specimens, an andalusite content of 25% (i.e., formulation D) is selected as the optimal experimental condition.

